Our Solution
We propose a self-healing concrete system that actively detects and repairs structural damage before it becomes critical, our technology integrates advanced sensors, nanobots, and a nanoparticle-based repair coating to continuously monitor stress, cracks, and internal changes within infrastructure.

“Use this space to share reviews from customers about the products or services offered.”

01. Initial Setup of Sensor + Nanobots
The initial setup consists of the microscopic nanobots embedding themselves within the concrete.
AE and FBG sensors will be manually installed conservatively on the surface of the concrete to communicate with millions of nanobots simultaneously.
(Utilize arrows to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

02. Crack Forms Within Concrete
A crack is formed within the deeper layers of the concrete structure.
This could be through repeated environmental erosion, prolonged stress load, or poor structural strength.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. Wave Reaches AE Sensors
The crack releases acoustic waves (high-frequency stress waves), which are detected by the AE and FBG sensors.
AE sensors regulate quick, instantaneous pressure gradient changes while FBG sensors monitor long-term stress build up.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

04. AE Sensor Signals Nanobots
The AE sensors determine the location of the crack through multilateration and communicate this information to the nanobots.
The nanobots are now aware of where the crack is within the volume of concrete.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

05. Nanoparticle Release
The nanobots release our novel repair-based nanoparticles into the concrete pore fluid.
These nanoparticles will have a diameter of 0.1 nm, allowing them to move through pores ranging from 0.5 nanometers to 5 micrometers.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

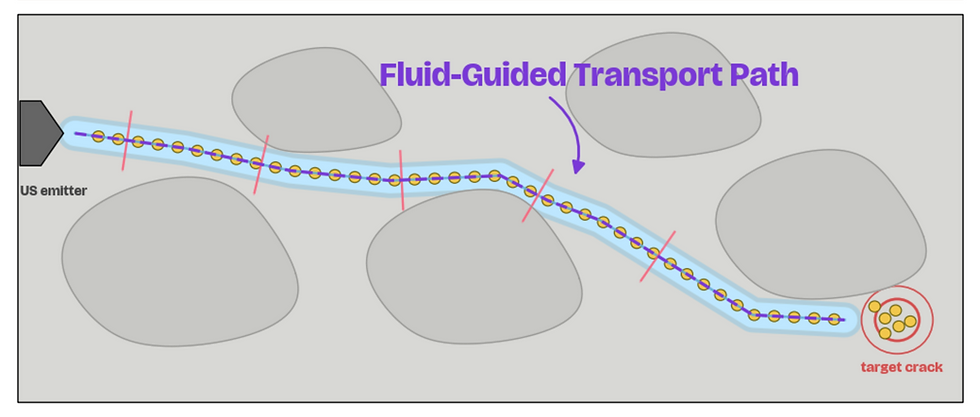
06. Ultrasonic Guidance to Crack
Our nanobot uses the ultrasonic emitters to release waves that guide the particles to the crack location.
The wave will purposefully morph its path in order to create fluidic flow within pore fluid that carries the particles.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
Video
When damage is detected, nanobots embedded on the surface are triggered to release specialized, repair-based nanomaterials that fill cracks and restore structural integrity in real time. Compared to current solutions that rely on delayed manual inspections and costly repairs, this system is low-maintenance.
When damage is detected, nanobots embedded on the surface are triggered to release specialized, repair-based nanomaterials that fill cracks and restore structural integrity in real time. Compared to current solutions that rely on delayed manual inspections and costly repairs, this system is low-maintenance.
(Swipe left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
When damage is detected, nanobots embedded on the surface are triggered to release specialized, repair-based nanomaterials that fill cracks and restore structural integrity in real time. Compared to current solutions that rely on delayed manual inspections and costly repairs, this system is low-maintenance.
Nanobot
The nanobot is a stationary, surface-embedded device composed of four main components: a micro-scale power source (battery), ultrasonic sensing emitters to guide particle movement, an internal storage unit that holds healing nanoparticles, and a controlled nozzle-release system that deploys them when damage is detected.
Together, these components allow the nanobot to sense structural failure and initiate a targeted repair response.
_gif.gif)
(Animation created by student researchers, Fusion360, 2026)
(Schematic diagram of nanobot model created by student researchers, Fusion360, 2026)





(3D printing the nanobot model)

(Images of 3D printed nanobot model)
Pore fluid in concrete is the liquid inside tiny pores, made of water and dissolved ions; although many people think concrete is completely solid, pore fluid typically makes up about 15–25% of its volume and strongly influences chemical reactions and corrosion [12], [13].

(Fluid Transport and Trapping Mechanisms in Porous Concrete Microstructure, SOURCE)
Transport Mechanism
Pore Fluid
Ultrasound waves are high-frequency vibrations that create pressure changes in a fluid. These changes can produce effects like acoustic streaming, which drives fluid flow and mixing [14].

(Ultrasound-Induced Acoustic Streaming and Resulting Fluid Transport in a Confined Medium, SOURCE)
Nanobot Targeted Transport
Our nanobot takes advantage of this pore fluid in between concrete particles, using ultrasonic waves to direct the fluid flow to the target crack. The released nanoparticles will then be carried by this fluid, through capillary networks, to the crack site.

“Use this space to share reviews from customers about the products or services offered.”

01. Nanobot Ultrasonic Emission
The nanobot releases waves from the ultrasonic emitters that drive the nanoparticles out from the release nozzle.
As explained previously, the waves will cause fluid motion within the particles and their surroundings, causing motion.
(Utilize arrows to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
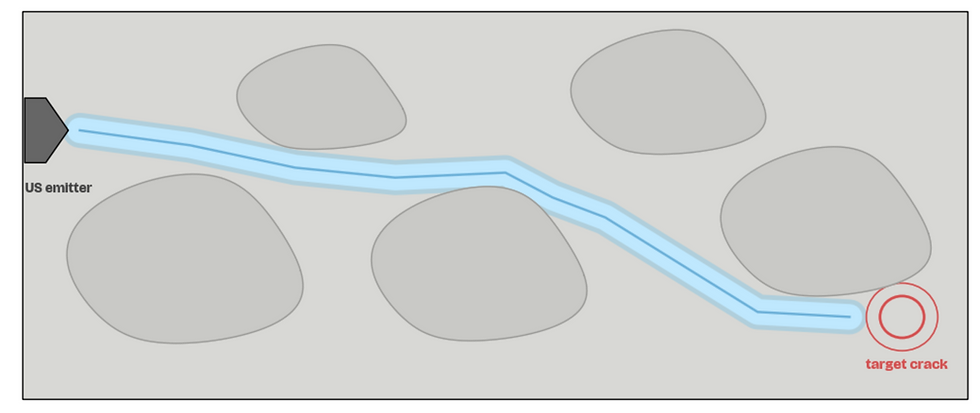
02. Crack Forms Within Concrete
A crack is formed within the deeper layers of the concrete structure.
This could be through repeated environmental erosion, prolonged stress load, or poor structural strength.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. Acoustic Wave Reaches AE Sensors
The crack releases acoustic waves (high-frequency stress waves), which are detected by the AE and FBG sensors.
AE sensors regulate quick, instantaneous pressure gradient changes while FBG sensors monitor long-term stress build up.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
04. AE Sensor Signals Nanobots
The AE sensors determine the location of the crack through multilateration and communicate this information to the nanobots.
The nanobots are now aware of where the crack is within the volume of concrete.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
(Swipe left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
The nanobot releases waves from the ultrasonic emitters that drive the nanoparticles out from the release nozzle.
As explained previously, the waves will cause fluid motion within the particles and their surroundings, causing motion.
01. Nanobot Ultrasonic Emission
Nanoparticle
What is C-S-H Gel?
Our nanoparticles will exploit the ion-rich pore fluid surrounding them and absorb free-flowing ions to artificially create additional C-S-H gel within the concrete at the crack site, repairing any damage.
Calcium Silicate Hydrate (C–S–H) gel is the primary binding phase in concrete and is responsible for the majority of its mechanical strength and long-term durability [12], [13].
It forms during the hydration of Portland cement, where calcium silicates react with water to produce a cohesive, gel-like structure that binds aggregates together [12].
%20(1).jpg)
.jpg)
.jpg)


(Layered Atomic Structure of Calcium–Silicate–Hydrate (C–S–H) Gel, SOURCE)
(Images of 3D printed nanoparticle model; swipe trackpad to view pictures)
(Schematic diagram of nanoparticle model created by student researchers, Fusion360, 2026)





Calcium Silicate Core
The calcium–silicate core serves as the structural backbone of the nanoparticle, providing mechanical stability and chemical compatibility with the surrounding cementitious matrix. Its composition allows it to remain stable.
BaTiO3 Piezoelectric Layer
The barium titanate (BaTiO₃) layer introduces a piezoelectric functionality, meaning it can generate an electric potential when subjected to mechanical stress [15]. In the context of the nanoparticle, stress from loading or crack propagation can activate this layer, producing localized electric fields. These fields can influence ion distribution through electrostatic interactions.
Catalytic Layer
This layer serves as the active interface between the nanoparticle and the surrounding pore fluid, providing nucleation sites that accelerate the formation of calcium–silicate–hydrate (C–S–H) gel. In cement chemistry, nucleation refers to the process by which dissolved ions organize into stable clusters, initiating solid-phase formation [13], [14]. Once Ca²⁺, OH⁻, and dissolved silicate species enter the catalytic layer, nucleation occurs and these clusters grow into C–S–H gel.
Alkali Resistant Shell
The outer shell is designed to withstand the highly alkaline environment of concrete (pH ~12–13), where the pore solution contains ions such as Ca²⁺ and OH⁻ that continuously diffuse through the material [12], [13]. Controlled porosity enables selective transport of these ions and water into the inner catalytic layer, allowing for localized chemical reactions to occur [15].
(Interactive diagram of sections in the nanoparticle model. Hover over gray dots to read a description of the funciton for each layer!)


“Use this space to share reviews from customers about the products or services offered.”

01. Nanoparticle Enters Crack
Zoomed in view of the nanoparticle first entering the target crack after being moved to the location through ultrasonic waves.
The following slides show the interaction of only one of these particles. On a larger scale, significant results are obtained in terms of repair.
(Swipe left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

02. Structure of Nanoparticle
Pore fluid contains Ca2+, OH-, and silicate species. The nanoparticle is surrounded by these ions.
Furthermore, the nanoparticle consists of four layers: an alkali-resistant shell, a catalytic layer, a piezoelectric interlayer, and a calcium silicate core.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. BaTiO3 Interlayer Polarizes
Once the nanoparticle reaches the crack, the pressure generated by the continuous use of the structure's piezoelectricity propagates an electrical field within the interlayer.
This causes one side of the nanoparticle to be positively charged and the other negatively charged.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

04. Surrounding Ions are Drawn Inward
The generated electrical field draws charged particles from the pore fluid to form C-S-H gel.
More specifically, the particles pass through the pores of the shell and bond to sites on the catalytic layer, allowing for the formation of the
C-S-H gel.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
Zoomed in view of the nanoparticle first entering the target crack after being moved to the location through ultrasonic waves.
The following slides show the interaction of only one of these particles. On a larger scale, significant results are obtained in terms of repair.
01. Nanoparticle Enters Crack

“Use this space to share reviews from customers about the products or services offered.”

01. Nanoparticle Enters Crack
Zoomed in view of the nanoparticle first entering the target crack after being moved to the location through ultrasonic waves.
The following slides show the interaction of only one of these particles. On a larger scale, significant results are obtained in terms of repair.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

02. Structure of Nanoparticle
Pore fluid contains Ca2+, OH-, and silicate species. The nanoparticle is surrounded by these ions.
Furthermore, the nanoparticle consists of four layers: an alkali-resistant shell, a catalytic layer, a piezoelectric interlayer, and a calcium silicate core.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. BaTiO3 Interlayer Polarizes
Once the nanoparticle reaches the crack, the pressure generated by the continuous use of the structure's piezoelectricity propagates an electrical field within the interlayer.
This causes one side of the nanoparticle to be positively charged and the other negatively charged.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

04. Surrounding Ions are Drawn Inward
The generated electrical field draws charged particles from the pore fluid to form C-S-H gel.
More specifically, the particles pass through the pores of the shell and bond to sites on the catalytic layer, allowing for the formation of the
C-S-H gel.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
Catalytic Layer Interactions
Following ultrasonic-guided transport, ions such as Ca²⁺, OH⁻, and dissolved silicate species accumulate at the catalytic layer, where they undergo nucleation and growth into calcium–silicate–hydrate (C–S–H) gel. This process transforms transported chemical species into a solid binding phase, initiating localized self-healing within the crack [13], [15].

“Use this space to share reviews from customers about the products or services offered.”

01. Ca²⁺ Ion
Approach Method
Calcium ions (Ca²⁺) from the pore fluid diffuse toward negatively charged oxygen sites on the silicate surface.
Electrostatic attraction draws these ions into proximity with the catalytic layer, initiating interaction.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

02. Surface Complex Formation
The Ca²⁺ ions coordinate with oxygen atoms on the silicate structure, forming a surface complex.
This stabilizes the ions at the interface and prepares the site for further chemical reactions.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. Nucleation of
C–S–H
Additional ions aggregate at the surface, forming a stable nucleus of calcium–silicate–hydrate (C–S–H).
This marks the transition from dissolved species to a solid-phase structure.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

04. Initial Layer Formation
The nucleus expands as more Ca²⁺ and silicate species attach, creating a small, organized C–S–H layer.
This early structure begins to resemble the layered morphology of mature C–S–H.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

05. Network Growth of C-S-H
Silicate chains extend and are bridged by calcium ions, forming a larger interconnected network.
The structure grows laterally along the surface, increasing coverage and stability.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

06. Extend C-S-H Gel Formation
The network continues to thicken into a continuous, hydrated C–S–H gel with a layered structure.
This gel fills space and binds the surrounding material, contributing to mechanical strength and crack sealing.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

“Use this space to share reviews from customers about the products or services offered.”

01. Ca²⁺ Ion
Approach Method
Calcium ions (Ca²⁺) from the pore fluid diffuse toward negatively charged oxygen sites on the silicate surface.
Electrostatic attraction draws these ions into proximity with the catalytic layer, initiating interaction.
(Swipe left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

02. Surface Complex Formation
The Ca²⁺ ions coordinate with oxygen atoms on the silicate structure, forming a surface complex.
This stabilizes the ions at the interface and prepares the site for further chemical reactions.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

03. Nucleation of
C–S–H
Additional ions aggregate at the surface, forming a stable nucleus of calcium–silicate–hydrate (C–S–H).
This marks the transition from dissolved species to a solid-phase structure.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

04. Initial Layer Formation
The nucleus expands as more Ca²⁺ and silicate species attach, creating a small, organized C–S–H layer.
This early structure begins to resemble the layered morphology of mature C–S–H.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

05. Network Growth of C-S-H
Silicate chains extend and are bridged by calcium ions, forming a larger interconnected network.
The structure grows laterally along the surface, increasing coverage and stability.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)

06. Extend C-S-H Gel Formation
The network continues to thicken into a continuous, hydrated C–S–H gel with a layered structure.
This gel fills space and binds the surrounding material, contributing to mechanical strength and crack sealing.
(Utilize arrows in the top left to switch between pictures in the slideshow. Image created by student researchers, Google Collab, 2026)
01. Ca²⁺ Ion
Approach Method
Calcium ions (Ca²⁺) from the pore fluid diffuse toward negatively charged oxygen sites on the silicate surface.
Electrostatic attraction draws these ions into proximity with the catalytic layer, initiating interaction.